14-2 Fluids at Rest#
Prompts
How does pressure vary with depth in a liquid? With altitude in the atmosphere? What is the physical reason?
Derive or explain \(p = p_0 + \rho g h\). What does each term represent?
Why is pressure the same at all points at the same level in a fluid at rest?
What is gauge pressure? How does it differ from absolute (total) pressure? When would you use each?
A scuba diver ascends without exhaling. Why is this dangerous? Relate to pressure difference between lungs and surroundings.
Lecture Notes#
Overview#
Hydrostatic pressure: pressure in a fluid at rest varies with vertical position—increases with depth, decreases with altitude.
Key relation: \(p = p_0 + \rho g h\) for depth \(h\) below a reference level.
Energy view: \(\rho g h\) is the gravitational potential energy per unit volume at depth \(h\); the pressure increment reflects the weight of the column above.
Same level → same pressure (same gravitational PE density). Absolute vs gauge pressure.
Quantity |
Meaning |
|---|---|
Absolute pressure \(p\) |
Total pressure at a point |
Gauge pressure \(p_g\) |
\(p - p_0\) (excess over atmospheric) |
Hydrostatic pressure#
Consider a fluid at rest. Take a vertical \(y\) axis with positive upward, origin at a reference level (e.g., liquid surface).
For two levels at \(y_1\) and \(y_2\):
\(\rho\): fluid density, \(g\): free-fall acceleration
Physical meaning: The extra pressure at level 2 comes from the weight of the fluid column between the two levels.
Depth below surface: If level 1 is the surface (\(p_1 = p_0\), atmospheric) and level 2 is at depth \(h\) below it (\(y_2 = -h\)):
\(p\): pressure at depth \(h\)
\(p_0\): atmospheric pressure on the surface
\(\rho g h\): pressure due to the weight of the fluid above
Important
Pressure at a given depth depends only on depth, not on the horizontal size or shape of the container. Eq. (17) holds regardless of container shape.
Energy density conversion
From an energy perspective, the pressure at depth \(h\) exceeds the surface pressure by \(\rho g h\) because fluid descending to that depth releases gravitational potential energy (PE) \((\rho g h)\) per unit volume. That released PE is stored as pressure—the fluid is compressed by the weight above, so the pressure increment = the PE density converted. Same depth → same pressure → same amount of gravitational PE stored as pressure energy.
Atmosphere: Eq. (16) also applies above the surface with \(\rho = \rho_{\text{air}}\): pressure decreases with altitude.
Pressure at the same level#
In a fluid at rest, pressure is the same at all points on a horizontal level. If it were not, fluid would flow sideways to equalize it. Static equilibrium implies no flow → uniform pressure on each horizontal plane.
Example: U-tube manometer
A U-tube contains mercury (\(\rho_{\text{Hg}} = 13.6\;\text{g/cm}^3\)) on one side and water (\(\rho_w = 1.0\;\text{g/cm}^3\)) on the other. The two liquids meet at the bottom. At the same horizontal level in the mercury and water columns, pressure is equal: \(p_0 + \rho_w g h_w = p_0 + \rho_{\text{Hg}} g h_{\text{Hg}}\). So \(\rho_w h_w = \rho_{\text{Hg}} h_{\text{Hg}}\), and \(h_w / h_{\text{Hg}} = \rho_{\text{Hg}} / \rho_w \approx 13.6\). The water column is about 14× taller than the mercury column for the same pressure difference.
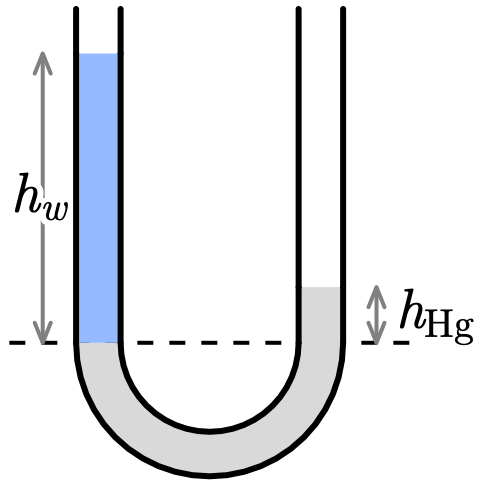
Fig. 4 U-tube manometer with water (height \(h_w\)) and mercury (height \(h_{\text{Hg}}\)).#
Absolute vs gauge pressure#
Absolute pressure (total pressure) \(p\): the actual pressure at a point. In Eq. (17), \(p\) is absolute.
Gauge pressure \(p_g = p - p_0\): the pressure in excess of atmospheric. For a liquid open to the air, \(p_g = \rho g h\).
Tire gauges, blood pressure cuffs, and many instruments read gauge pressure. Absolute pressure is needed for ideal gas law and thermodynamic calculations.
Diver danger
A diver who fills lungs at depth \(L\) and ascends without exhaling keeps lung pressure at \(p_0 + \rho g L\) while external pressure drops to \(p_0\). The pressure difference \(\Delta p = \rho g L\) can rupture lungs. Always exhale gradually during ascent.
Poll: Pressure at depth
Four containers of olive oil have the same depth \(h\) of oil. The containers have different shapes (wide, narrow, etc.). Rank the pressure at the bottom, greatest first.
(A) All the same
(B) Depends on container shape
(C) Wider container has greater pressure
(D) Narrower container has greater pressure

Fig. 5 Four containers with the same depth \(h\) of olive oil but different shapes.#
Summary#
Hydrostatic pressure: \(p_2 = p_1 + \rho g (y_1 - y_2)\); for depth \(h\): \(p = p_0 + \rho g h\)
Pressure depends on depth only, not container shape
Same horizontal level → same pressure
Gauge pressure \(p_g = p - p_0\); absolute \(p\) is total pressure

