14-1 Fluids, Density, and Pressure#
Prompts
What distinguishes a fluid from a solid? Why do we lump liquids and gases together as fluids?
Define density \(\rho\) and pressure \(p\). What are their SI units? How do they differ from mass and force?
For a fluid at rest, how does pressure at a point depend on the orientation of the surface? Is pressure a vector or scalar?
The air in a room has mass. How would you estimate the weight of the air using density and volume? What is the magnitude of atmospheric force on your head?
Compare gases and liquids: which is compressible? How does that affect their density under different pressures?
Lecture Notes#
Overview#
Fluids (liquids and gases) flow and conform to their container; solids have a rigid structure.
For fluids we use density \(\rho\) and pressure \(p\) instead of mass and force—these are field-like quantities that vary from point to point.
Density: mass per unit volume. Pressure: force per unit area (normal to the surface); pressure has units of energy per volume (Pa = J/m³).
Solid |
Fluid |
|
|---|---|---|
Structure |
Particles stay near fixed positions; often long-range positional order (crystals). |
Particles continuously rearrange; no long-range positional order. |
Shear |
Can sustain static shear stress. |
Cannot sustain static shear stress; flows under shear. |
Shape |
Has its own shape, largely independent of container. |
No fixed shape; takes container shape. |
Description |
Deformation of a body (displacement/strain). |
Pointwise fields (pressure, velocity, density). |
Why liquids and gases together?
Liquids and gases are often grouped together because they are both fluids. The key contrast is usually solid vs fluid, not liquid vs gas.
The deeper reason is structural. In a crystal solid, atoms are arranged in an ordered pattern that extends over long distances (long-range order), so knowing one atom’s position helps predict others far away. In liquids and gases, atoms may show only short-range local arrangement, but at large distances there is no fixed pattern (no long-range positional order). In this sense, both are “disordered” compared with a crystal.
In fact, in a phase diagram one can move continuously from liquid to gas by going around the critical point, without crossing a sharp phase boundary. This is not possible between solid and liquid, which are fundamentally different phases.
What is a fluid?#
A fluid is a substance that can flow. It conforms to the boundaries of its container because it cannot withstand shearing stress (a force tangential to its surface). It can, however, exert a force perpendicular to its surface.
Some materials (e.g., pitch) flow slowly but eventually conform—they are fluids.
Liquids and gases are both fluids; solids are not.
Density#
Density \(\rho\): mass per unit volume. At a point, take a small volume \(\Delta V\) and mass \(\Delta m\):
For a sample large compared to atomic dimensions (uniform density):
SI unit: kg/m³
Scalar (no direction)
Compressibility
Gases are readily compressible—density varies with pressure. Liquids are nearly incompressible—density changes little with pressure.
Poll: Mass of air in the room
A typical lecture room (e.g., 10 m × 8 m × 3 m) is filled with air at 1 atm. Which is the best estimate for the mass of air in the room? Use \(\rho_{\text{air}} \approx 1.2\;\text{kg/m}^3\).
(A) 3 kg
(B) 300 kg
(C) 3000 kg
(D) 30,000 kg
Pressure#
Pressure \(p\): force per unit area acting normal to a surface. For a small area \(\Delta A\) with normal force \(\Delta F\):
For a uniform force over a flat area \(A\):
SI unit: Pa (pascal) = N/m² = J/m³
Useful conversion: \(1\;\text{atm} = 1.01 \times 10^5\;\text{Pa} = 760\;\text{torr}\)
Pressure as a scalar
At a given point in a fluid at rest, pressure has the same value in all directions. Pressure is a scalar—the force on a sensor is a vector, but \(p = F/A\) uses only the magnitude of the normal force.
Poll: Pressure on your head
Atmospheric pressure at sea level is about \(1.0 \times 10^5\;\text{Pa}\). If the top of your head has area \(\approx 0.04\;\text{m}^2\), the magnitude of the atmosphere’s downward force on your head is closest to:
(A) 4 N
(B) 40 N
(C) 400 N
(D) 4000 N
Pressure as energy density
Since J = N m (work = force × distance), we have Pa = N/m² = J/m³. So pressure has units of energy per volume—just like density has units of mass per volume. Both \(\rho\) and \(p\) are per-volume scalars.
What kind of energy? Imagine a piston pushing a volume \(\Delta V\) of fluid through a pipe. Work done = \(F \cdot d = (pA) \cdot d = p \cdot \Delta V\). So pressure = work per unit volume—the potential energy a fluid has to expand and do work. High-pressure fluid is like a compressed spring: it carries stored capacity to do work even when stationary.
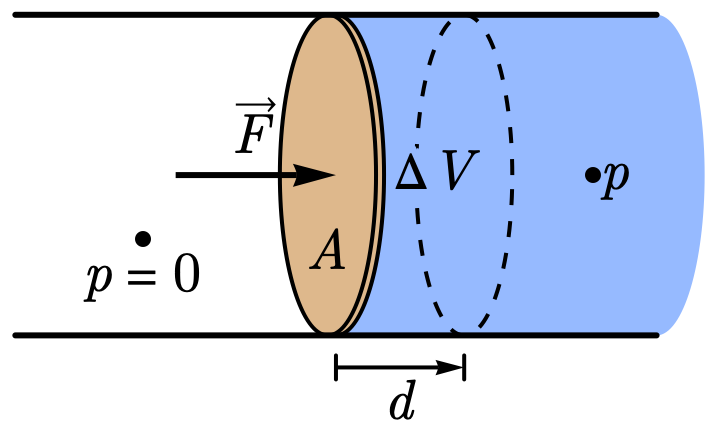
Fig. 2 Piston pushing fluid: force \(\vec{F}\) on area \(A\) displaces distance \(d\).#
Poll: What does pressure represent?
Pressure has units J/m³. In terms of energy, pressure best represents:
(A) Kinetic energy of moving molecules
(B) Work per unit volume the fluid can do when it expands
(C) Gravitational potential energy per unit volume
(D) Total energy stored in the fluid
Summary#
Fluid: flows; cannot withstand shearing stress; conforms to container.
Density \(\rho = m/V\): mass per unit volume (kg/m³); scalar.
Pressure \(p = F/A\): force per unit area normal to surface (Pa); scalar; same in all directions at a point.
Gases: compressible. Liquids: nearly incompressible.

